Guidance on the establishment of acceptable daily exposure limits (ADE) to support Risk-Based Manufacture of Pharmaceutical Products - ScienceDirect
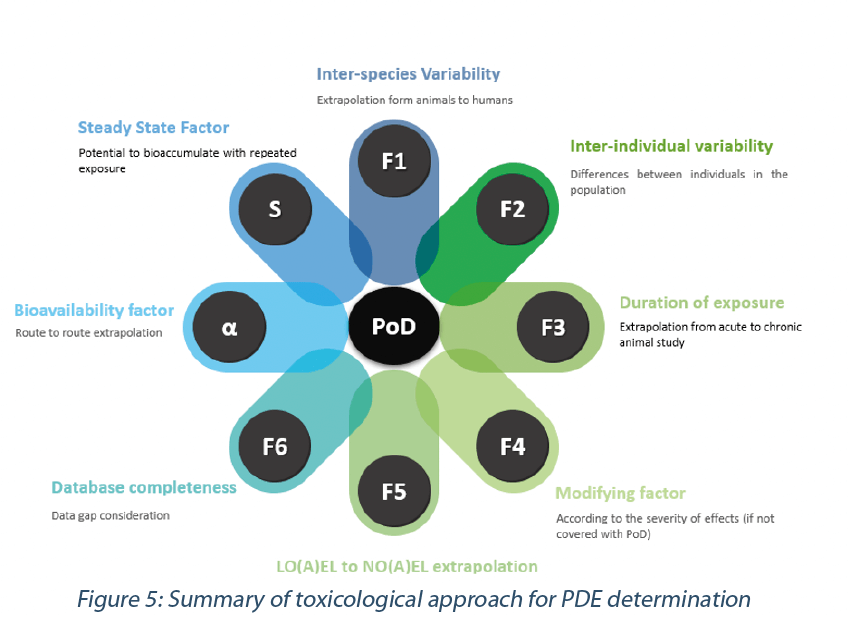
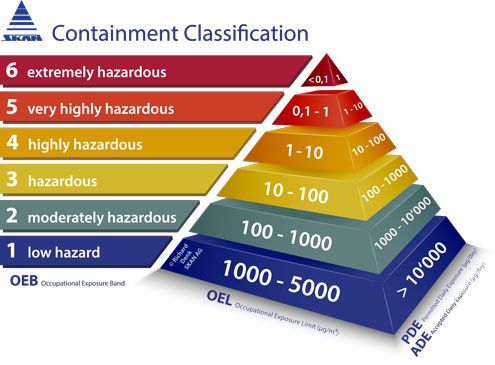
Toxicological approach to define the PDE for your cleaning validation process. - A3P - Pharmaceutical & Biotechnology Industry

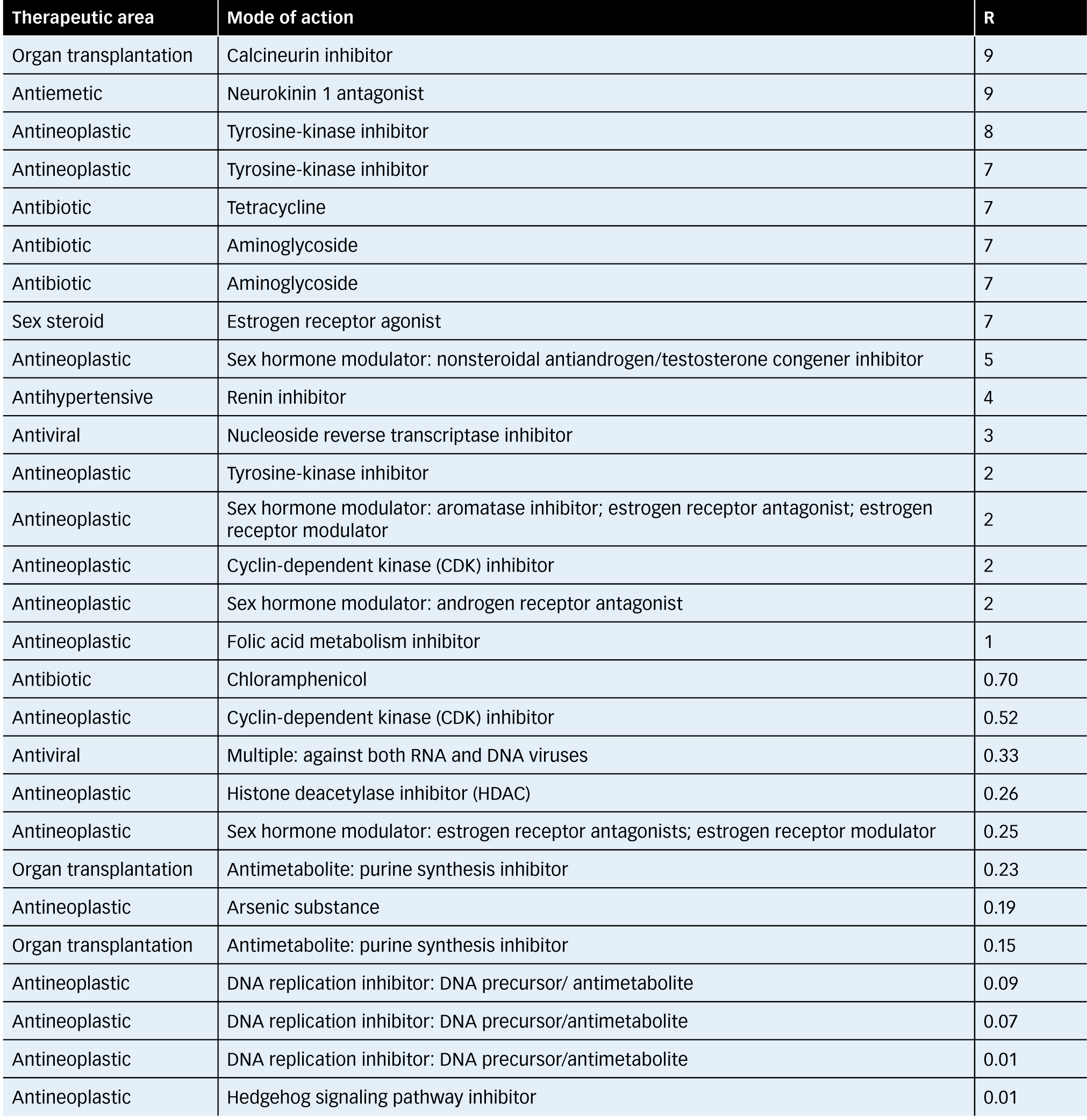
Table 2 from Use of the permitted daily exposure (PDE) concept for contaminants of intravitreal (IVT) drugs in multipurpose manufacturing facilities | Semantic Scholar

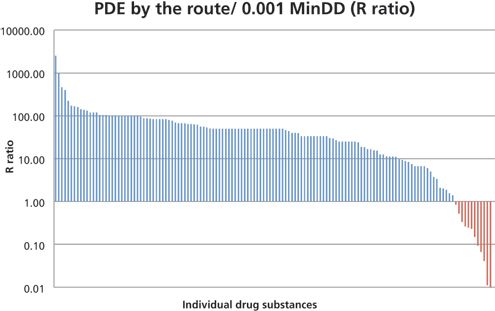
Permitted Daily Exposure (PDE) Certification Company | Acceptable Daily Exposure (ADE) PDI Value Derivation & Calculation

Use of the permitted daily exposure (PDE) concept for contaminants of intravitreal (IVT) drugs in multipurpose manufacturing facilities | Semantic Scholar

Guidance on the establishment of acceptable daily exposure limits (ADE) to support Risk-Based Manufacture of Pharmaceutical Products - ScienceDirect
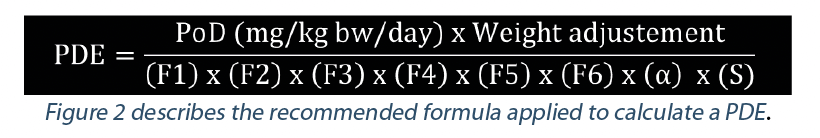
Toxicological approach to define the PDE for your cleaning validation process. - A3P - Pharmaceutical & Biotechnology Industry

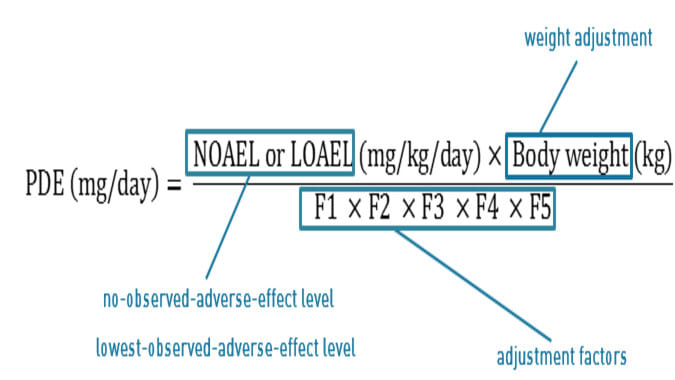
Calculation of a permitted daily exposure value for the solvent 2‐methyltetrahydrofuran | Semantic Scholar