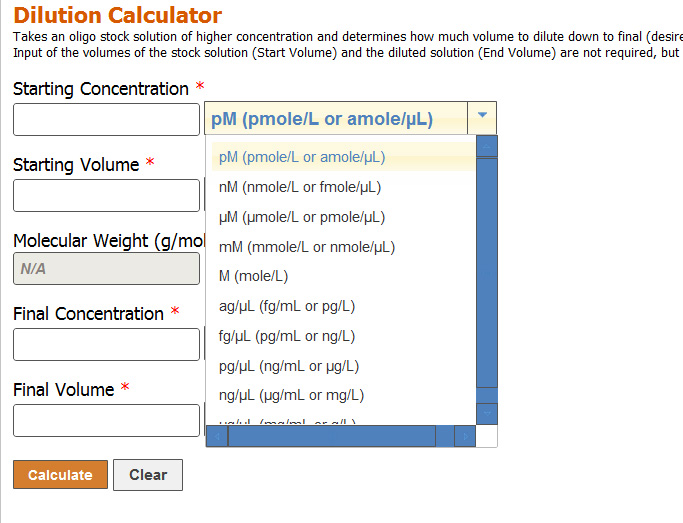
Question Video: Calculating the Molar Concentration of Mg(OH)₂ Using Data from a Titration Experiment | Nagwa

Hi can somebody tell me how to plug the anti log in the calculator I can't find it and also I don't understand the interpolation of this molar concentration equation : r/FE_Exam
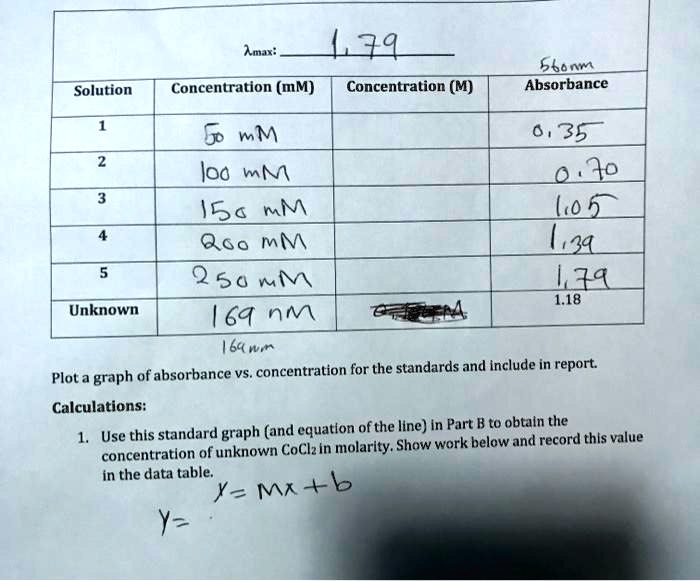
SOLVED: Amar: L39 Concentration (mM) Concentration (M) gbn4 Absorbance Solution mM lod mM 15a mM 060 mM 2 50 M 64 nm 64mm 0, 35 Jo Lo 43L LZA 1.18 Unknown Plot

How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com

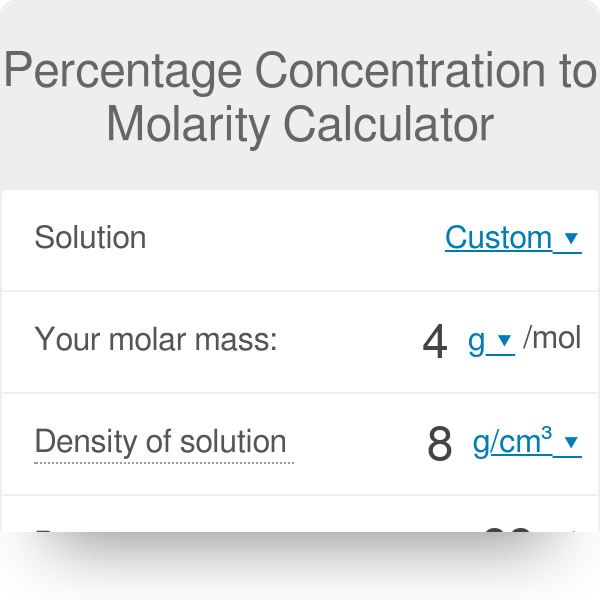


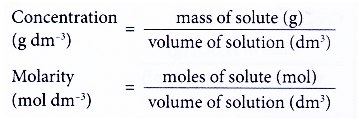
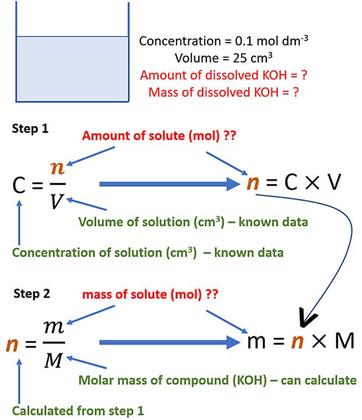
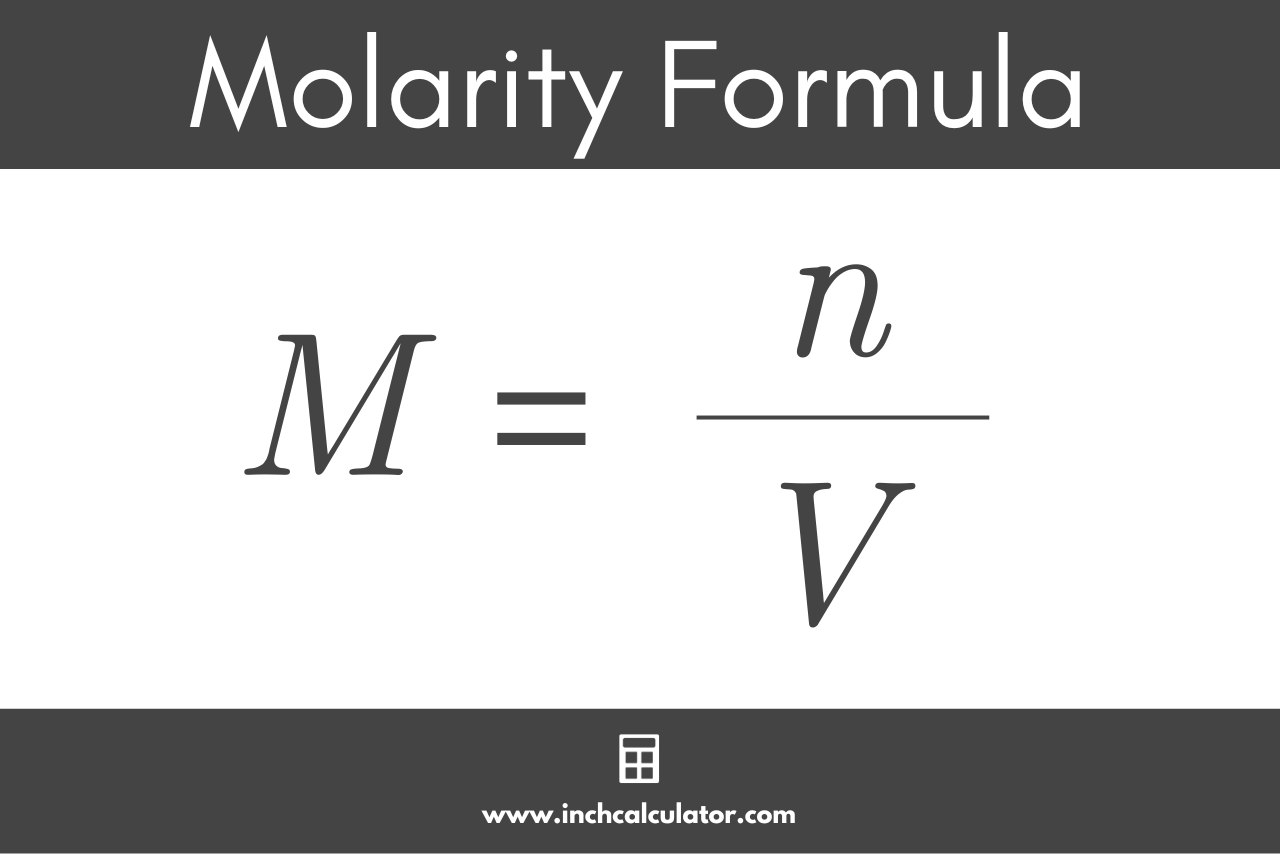

![Molarity Calculator [with Molar Formula] Molarity Calculator [with Molar Formula]](https://scrn-cdn.omnicalculator.com/chemistry/molarity@2.png)