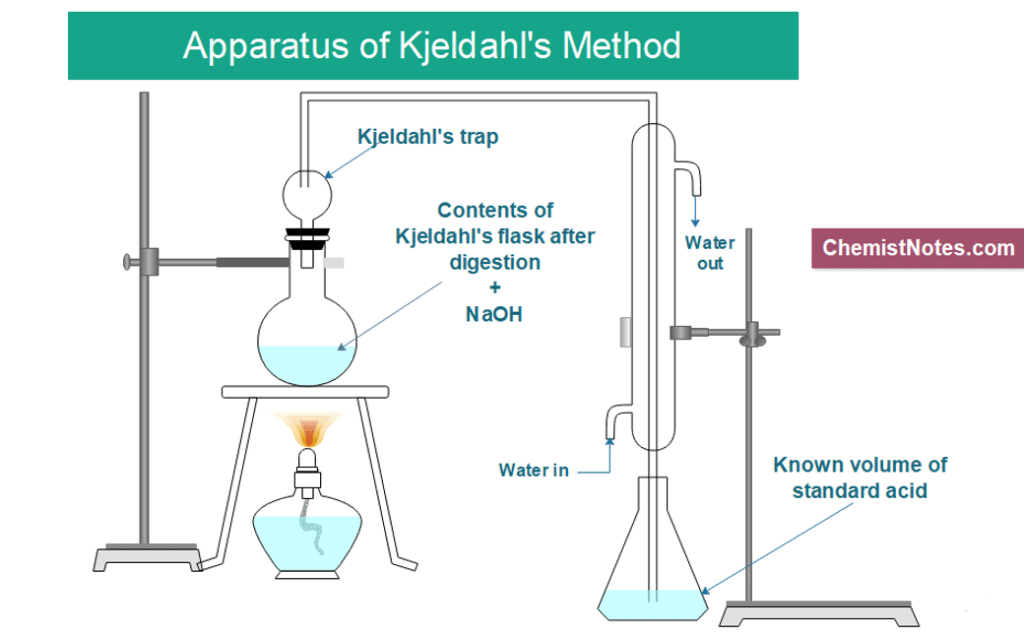
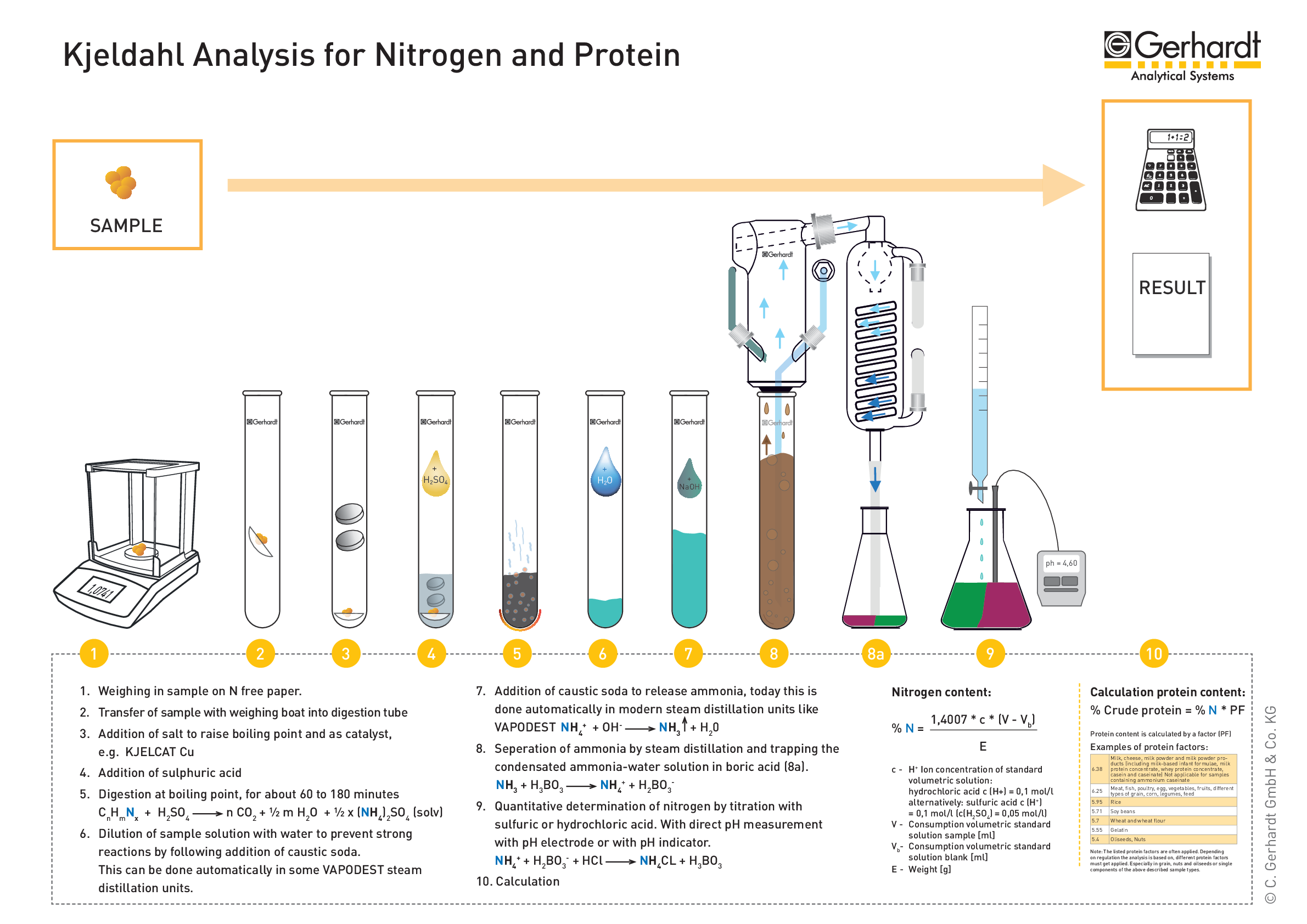
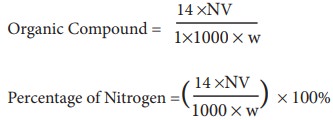In the Kjedahl's method for estimation of nitrogen present in soil sample, ammonia evolved from 0.75 g of sample neutralized 10ml. of 1M H2SO4 . The percentage of nitrogen in the soil is:

In the Kjedahl's method for estimation of nitrogen present in soil sample, ammonia evolved from 0.75 g of sample neutralized 10ml. of 1M H2SO4 . The percentage of nitrogen in the soil is:

7g of an organic compound containing N is treated by kjeldahl method and NH3 released is absorbed in 80ml semimolar - Chemistry - Solutions - 13533101 | Meritnation.com