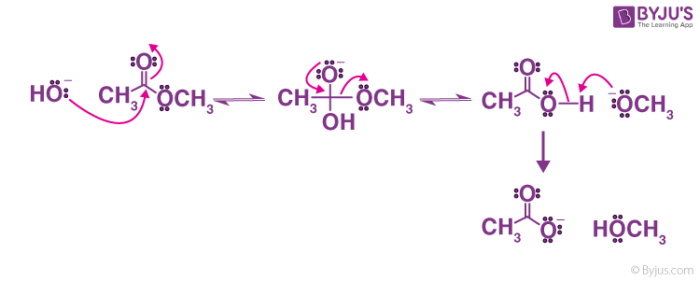
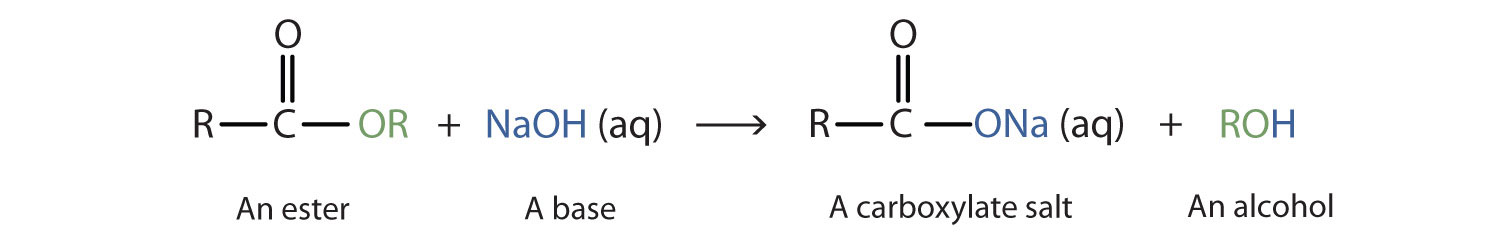
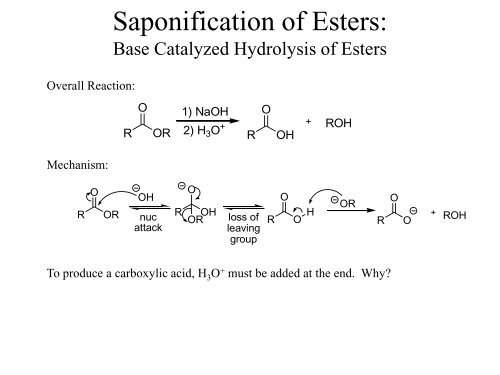
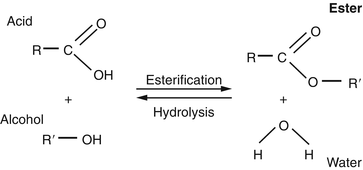
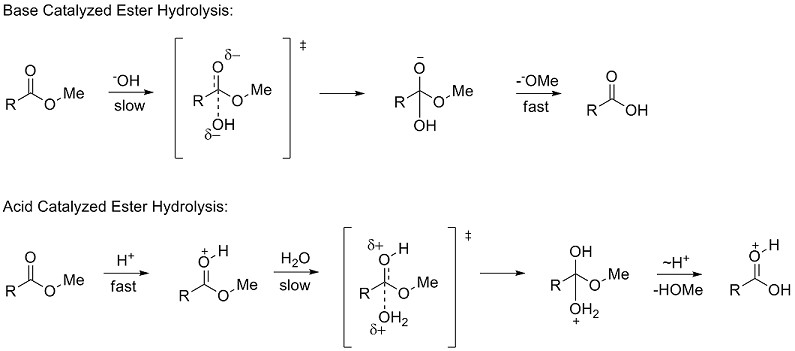
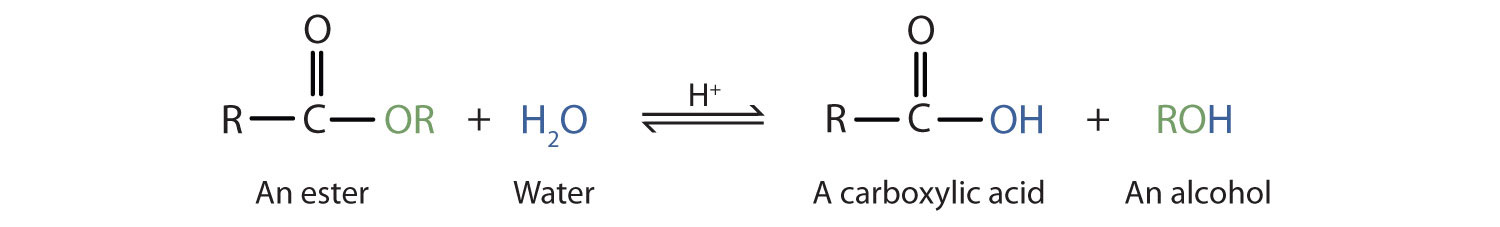
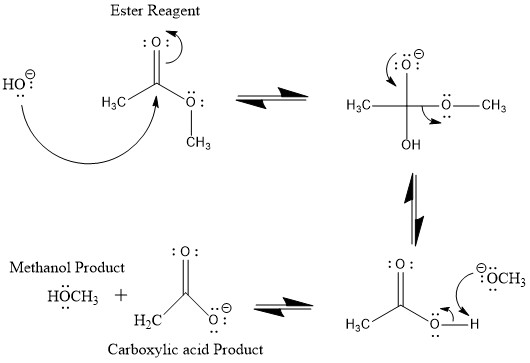
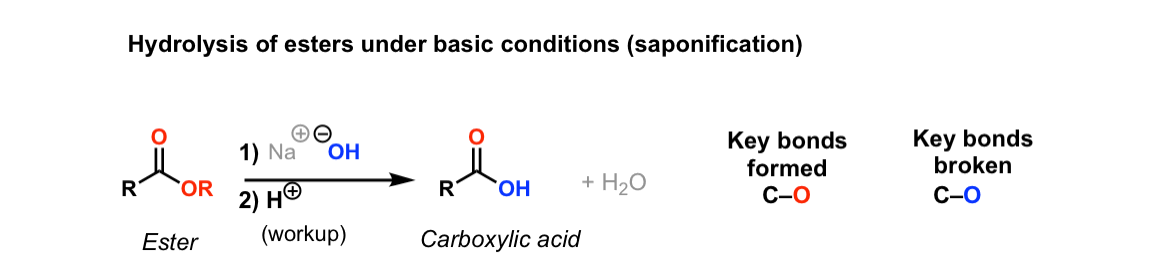
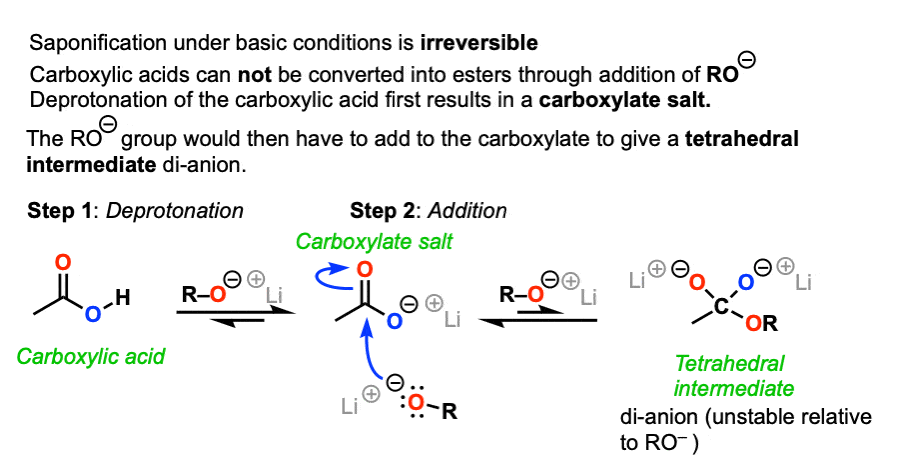
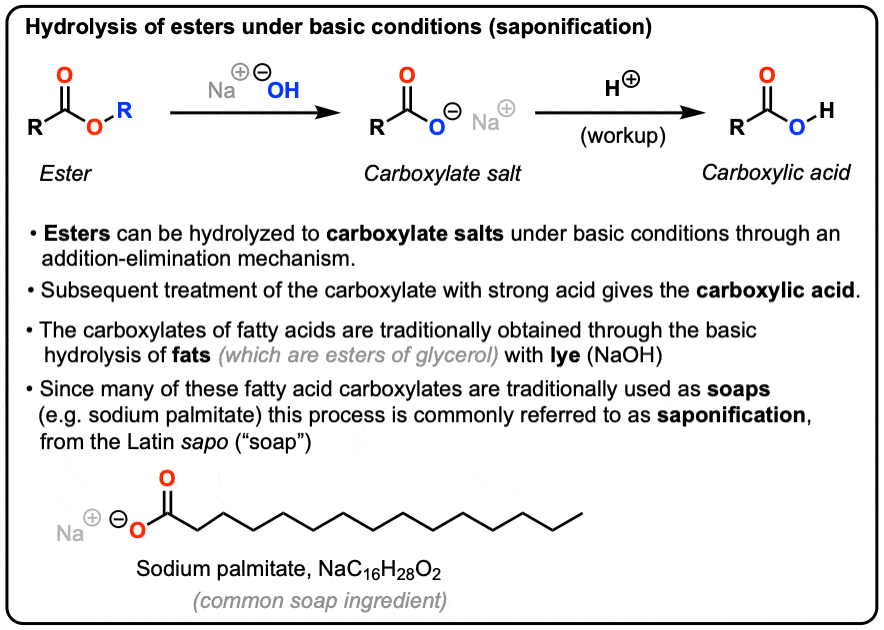
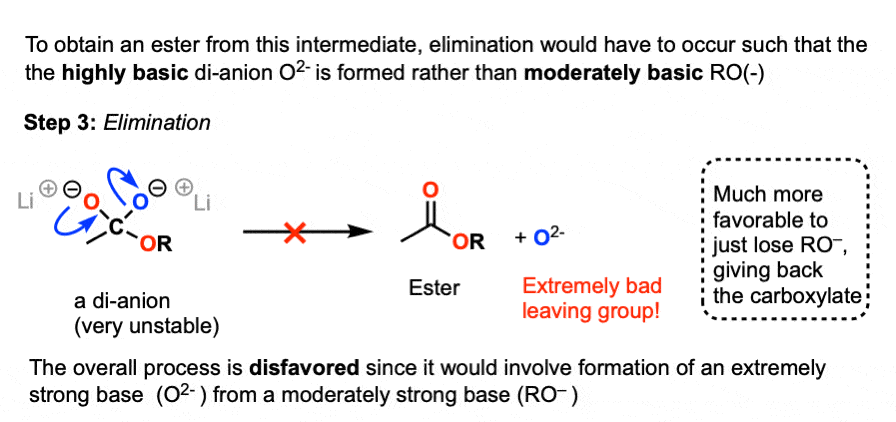
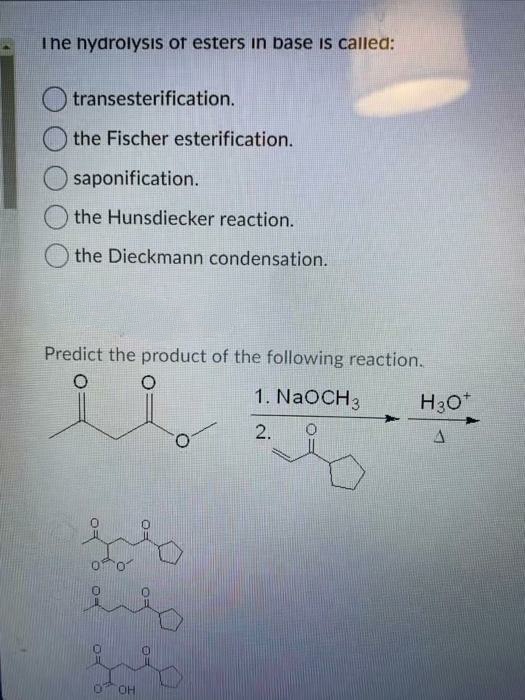
How can you convert a carboxylic acid into an ester? a. Heat with an alcohol and catalytic acid. b. Deprotonate with a base and react with an alcohol. c. Deprotonate with a

Revisiting the Mechanism of Neutral Hydrolysis of Esters: Water Autoionization Mechanisms with Acid or Base Initiation Pathways | The Journal of Physical Chemistry B