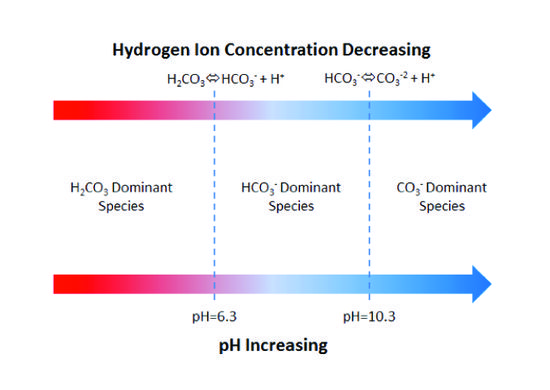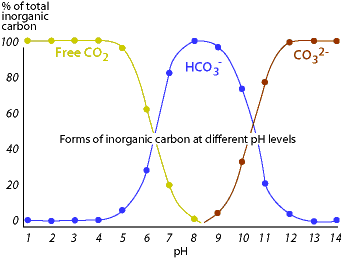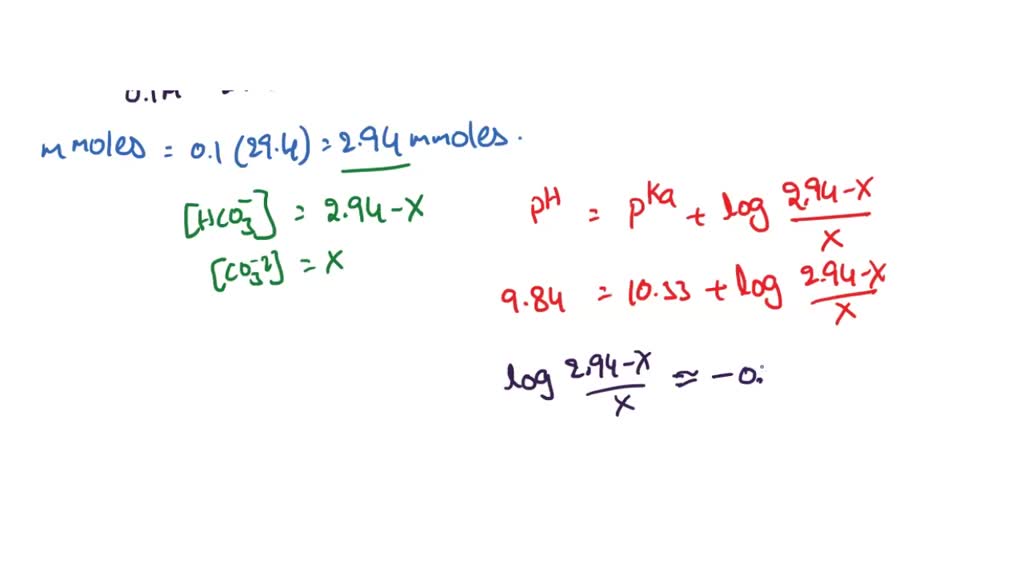
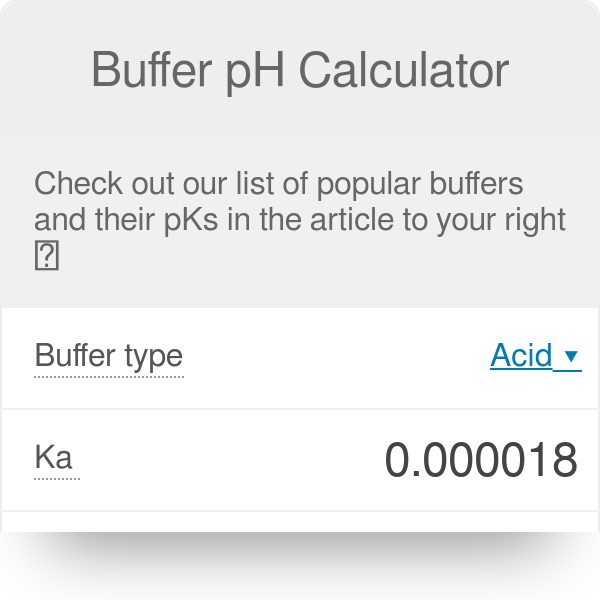
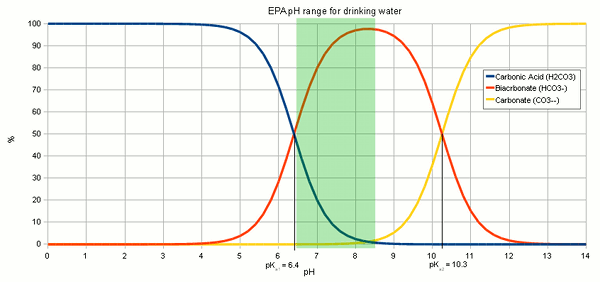
SOLVED: A bicarbonate buffer of pH 6.1 needs to be prepared. What would you add to 500 mL of 0.4 M sodium carbonate solution in order to make this buffer? (Hint: carbonic
![Calculating the equilibrium concentrations of carbonate, bicarbonate, carbonic acid and [H+] - YouTube Calculating the equilibrium concentrations of carbonate, bicarbonate, carbonic acid and [H+] - YouTube](https://i.ytimg.com/vi/KS8l5d7O-Yc/maxresdefault.jpg)
Calculating the equilibrium concentrations of carbonate, bicarbonate, carbonic acid and [H+] - YouTube
![PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/104ba10db168516a4ba7d20867d92f2a51b891e7/4-Table2-1.png)
PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar
![PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/104ba10db168516a4ba7d20867d92f2a51b891e7/3-Table1-1.png)







![PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aaa98c1f9de4c398b6a057bf4936ea76736034c7/1-Table1-1.png)